




 BRJ Medical
BRJ Medical










| Still deciding? Get samples of $ ! US$ 70/Piece Request Sample |


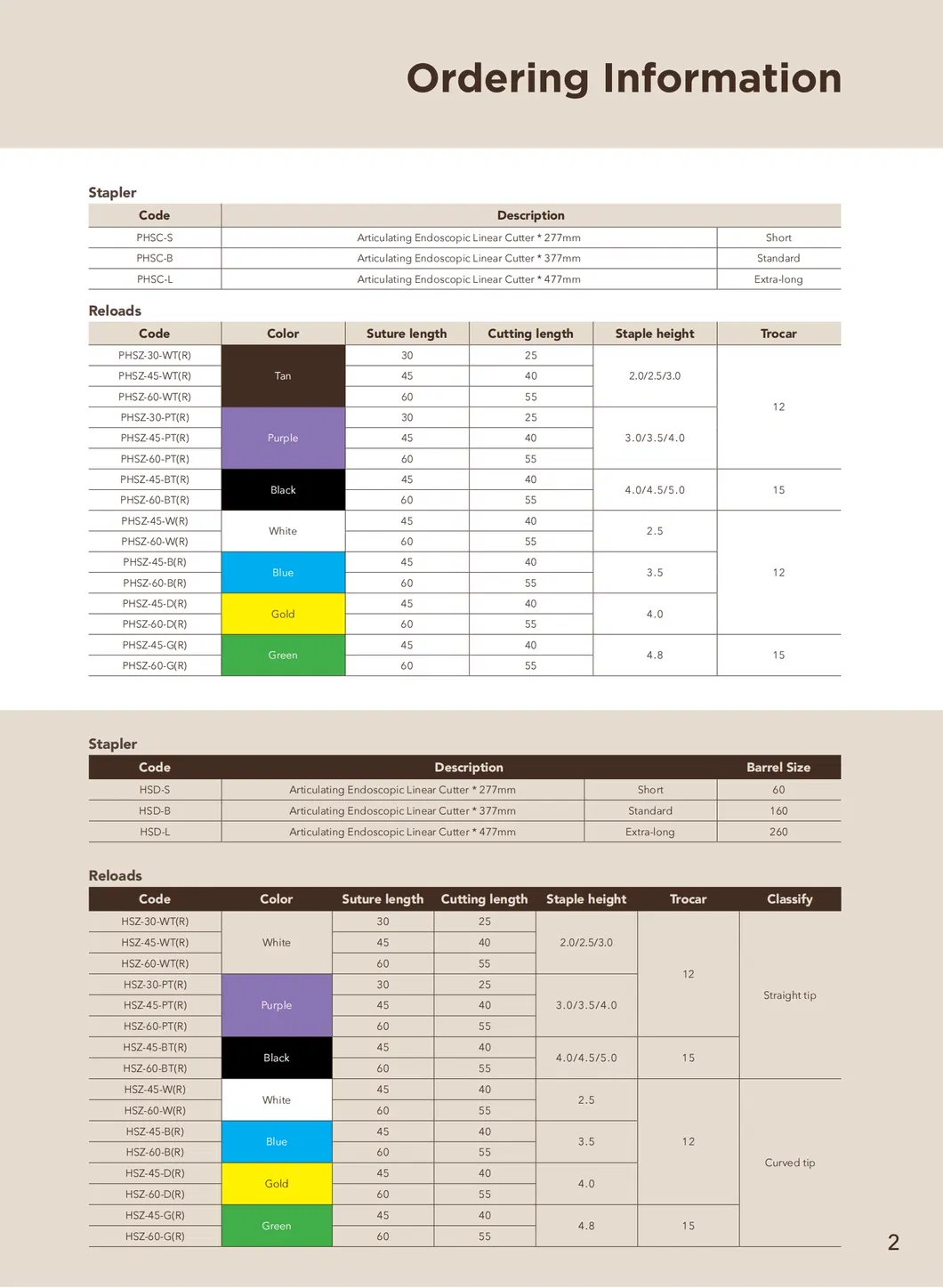
| Product Code | Model | Color | Suture Length ±5 | Cutting Length ±5 | Staple Height | Staple Width |
|---|---|---|---|---|---|---|
| 12029302 | HSZ-30-WT(R) | White | 30 | 25 | 2.5/3.0 | 3 |
| 12029303 | HSZ-30-PT(R) | Purple | 3.5/4.0 | |||
| 12029454 | HSZ-45-PT(R) | Purple | 45 | 40 | 3.0/3.5/4.0 | |
| 12029455 | HSZ-45-BT(R) | Black | 4.0/4.5/5.0 | |||
| 12029603 | HSZ-60-WT(R) | White | 60 | 55 | 2.0/2.5/3.0 | |
| 12029604 | HSZ-60-PT(R) | Purple | 3.0/3.5/4.0 | |||
| 12029605 | HSZ-60-BT(R) | Black | 4.0/4.5/5.0 |

As a national high-tech enterprise, the proportion of R&D investment to total sales has been over 8% for consecutive years. The facility operates strictly according to the ISO13485 quality system and CE MDD 93/42/EEC directive implementation.



Equipped with German-imported and American-imported machining centers and Japanese-imported automatic lathes, the testing center includes a biomechanical laboratory and a physical-chemical testing center for rigorous quality control.



The product is fully certified with CE, FDA, and ISO13485 standards, ensuring it meets international medical safety requirements.
Yes, we support 100% OEM and semi-finished supply to meet specific branding and market needs for global partners.
All endoscopic staplers undergo Ethylene Oxide (EO) sterilization to ensure they are safe for hospital and surgical use.
Yes, the universal handle is designed to be compatible with various reloading units, providing flexibility during different surgical procedures.
The device incorporates a safety interlock that prevents firing until the 'Pre-Fire' button is pressed. It also includes a cartridge lock-out to prevent the use of already fired reloads.
The facility has a robust production capacity of approximately 500,000 pieces per year, ensuring a steady supply for international markets.