




 BRJ Medical
BRJ Medical










| Customization: | Available |
|---|---|
| Noise: | 40-60db |
| Surface Treatment: | Baking Finish |
VHP sterilization is non-toxic and residue-free, degrading quickly into H2O and O2 after the process. This equipment ensures excellent material compatibility through a unique saturation control method, preventing liquefaction or condensation during sterilization.


A LOGA Program: Achieves set sterilization conditions generally set to a 6LOG reduction value.
B LOGB Program: A 12LOG overkill program for maximum sterilization security.
C Concentration Program: Controlled by concentration and time parameters based on specific development needs.
D Self-cleaning Procedure: Utilizes HEPA filtration for cavity cleaning, allowing material transfer without full sterilization cycle.
| Model | Inner Dimensions (mm) | External Dimensions (mm) | Power (kW) | Weight (kg) |
|---|---|---|---|---|
| TKSAGE HPB-216 | 600*600*600 | 1050*650*1800 | 2.0 | 280 |
| TKSAGE HPB-288 | 600*600*800 | 1050*650*2000 | 2.2 | 300 |
| TKSAGE HPB-512 | 800*800*800 | 1250*850*2000 | 2.3 | 350 |
| TKSAGE HPB-800 | 800*1000*1000 | 1250*1050*2200 | 2.6 | 380 |
| TKSAGE HPB-2688 | 1200*1400*1600 | 1850*1450*2150 | 4.0 | 600 |
Note: Custom configurations are available upon request for non-standard products.
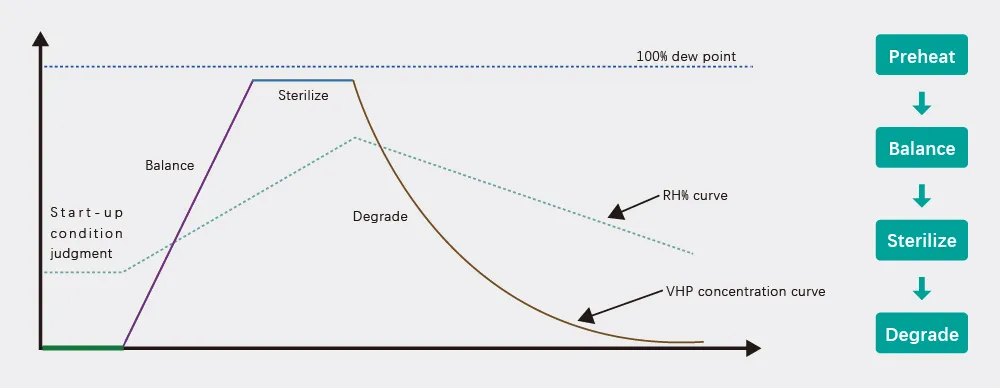
1. Preheat: Automatic adjustment of temperature and humidity conditions.
2. Balance: Self-balancing of VHP concentration and saturation.
3. Sterilize: Active sterilization until the set LOG value is reached.
4. Degrade: Exhaust stage for residue removal and degradation.
Our facilities include advanced CNC machinery, laser cutting, and fully automatic welding systems. Key production areas focus on filtration, sheet metal, and sterile assembly.







• ISO9001-2015 Quality Management System
• CE and UL International Certifications
• National High-Tech Enterprise Status
• Over 100 invention and utility patents



