 BRJ Medical
BRJ Medical










| Customization: | Available |
|---|---|
| Type: | Foley Catheter |
| Material: | Silicone |
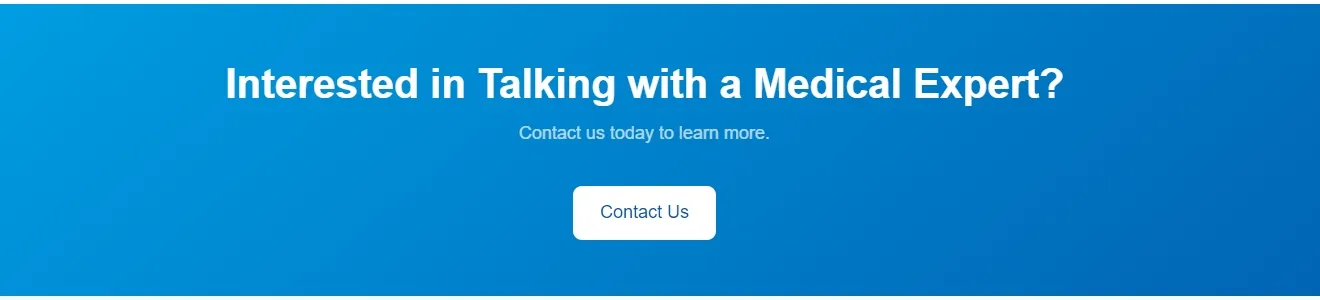
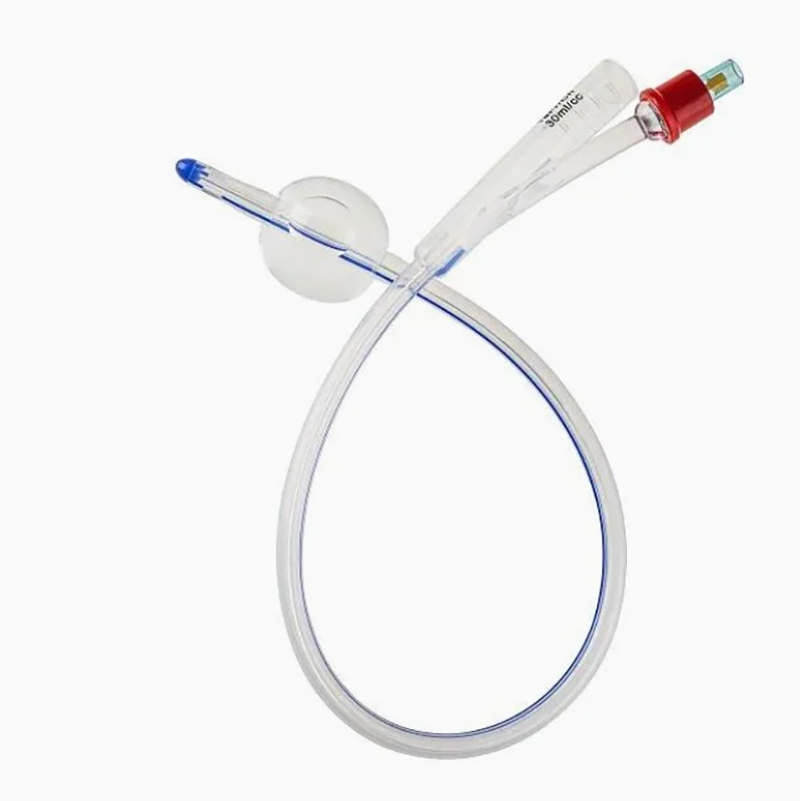
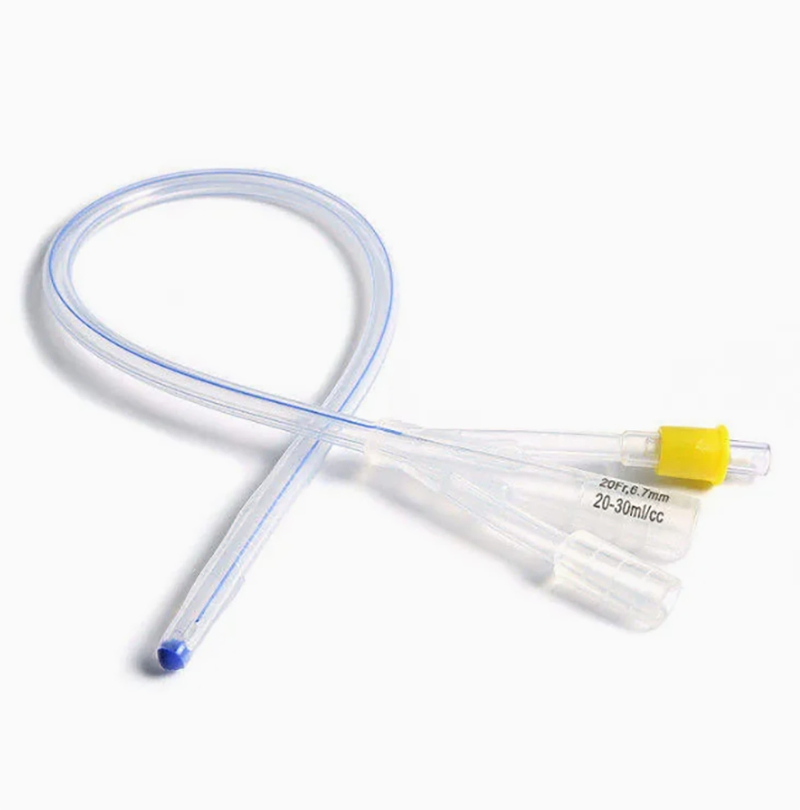
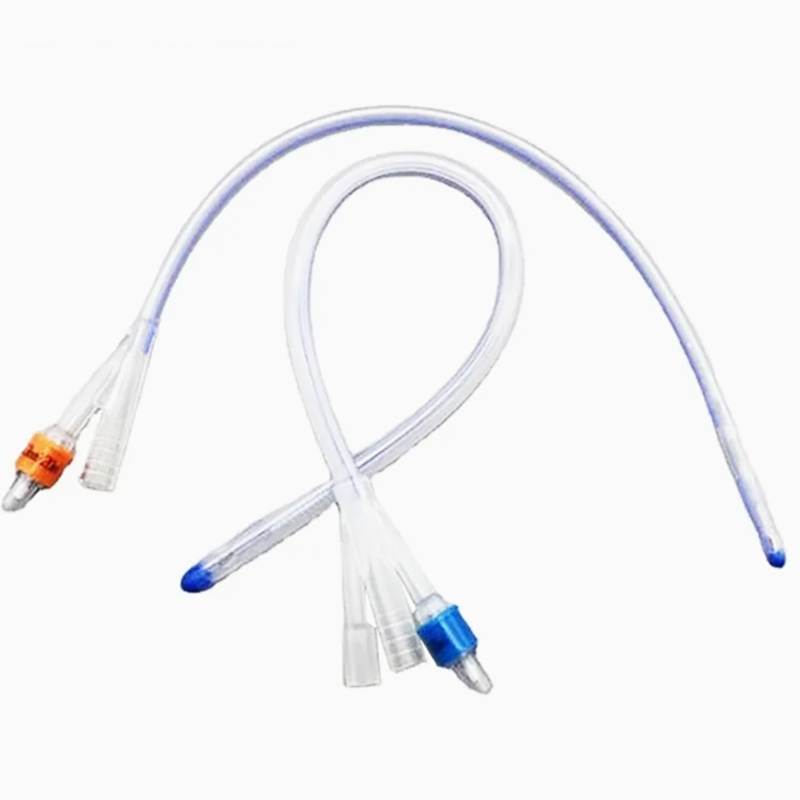
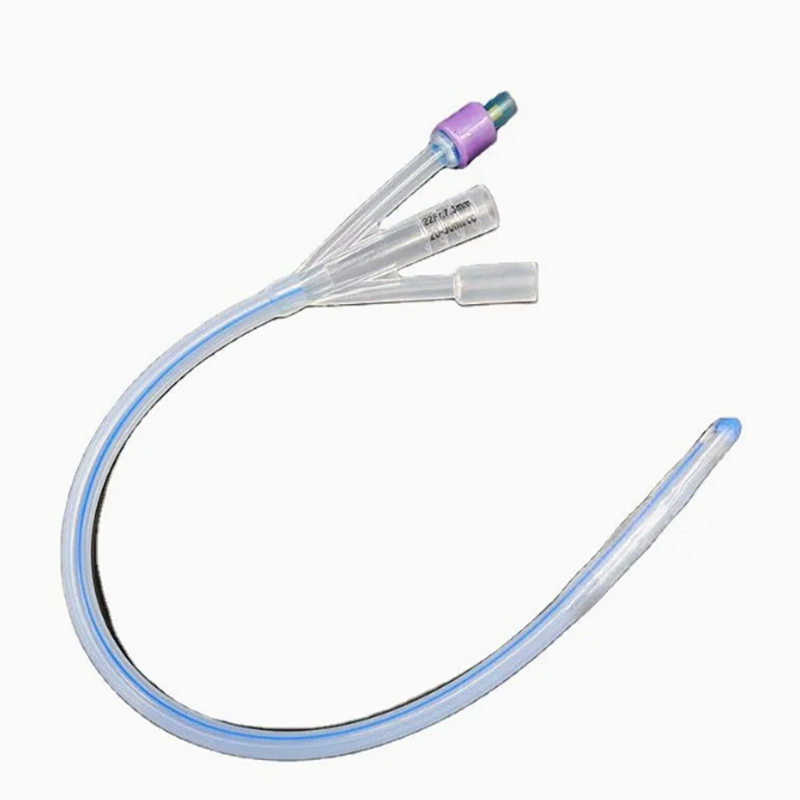
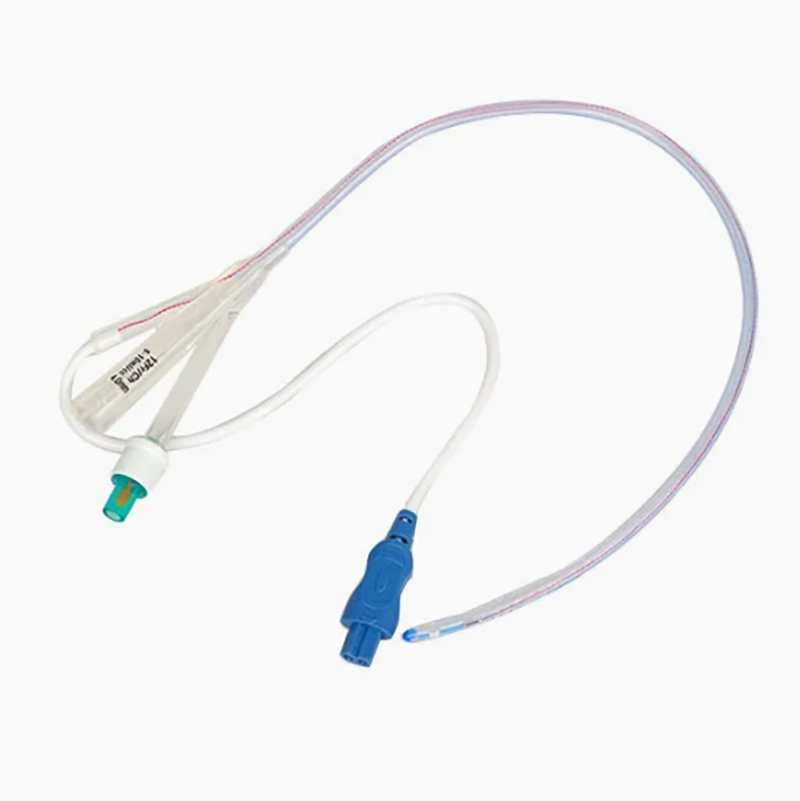
Silicone Foley Catheter
Sterile, single-use urinary catheter made of 100% medical-grade silicone for superior biocompatibility. Features a soft tip, large symmetric balloon (3-30ml) for secure bladder retention, and full-length radiopaque line for X-ray positioning. Color-coded connectors simplify size identification; available in 2/3-way variants. Smooth anti-encrustation surface and optional hydrophilic coating enhance comfort. EO-sterilized, used for routine clinical catheterization, drug administration, and flushing.
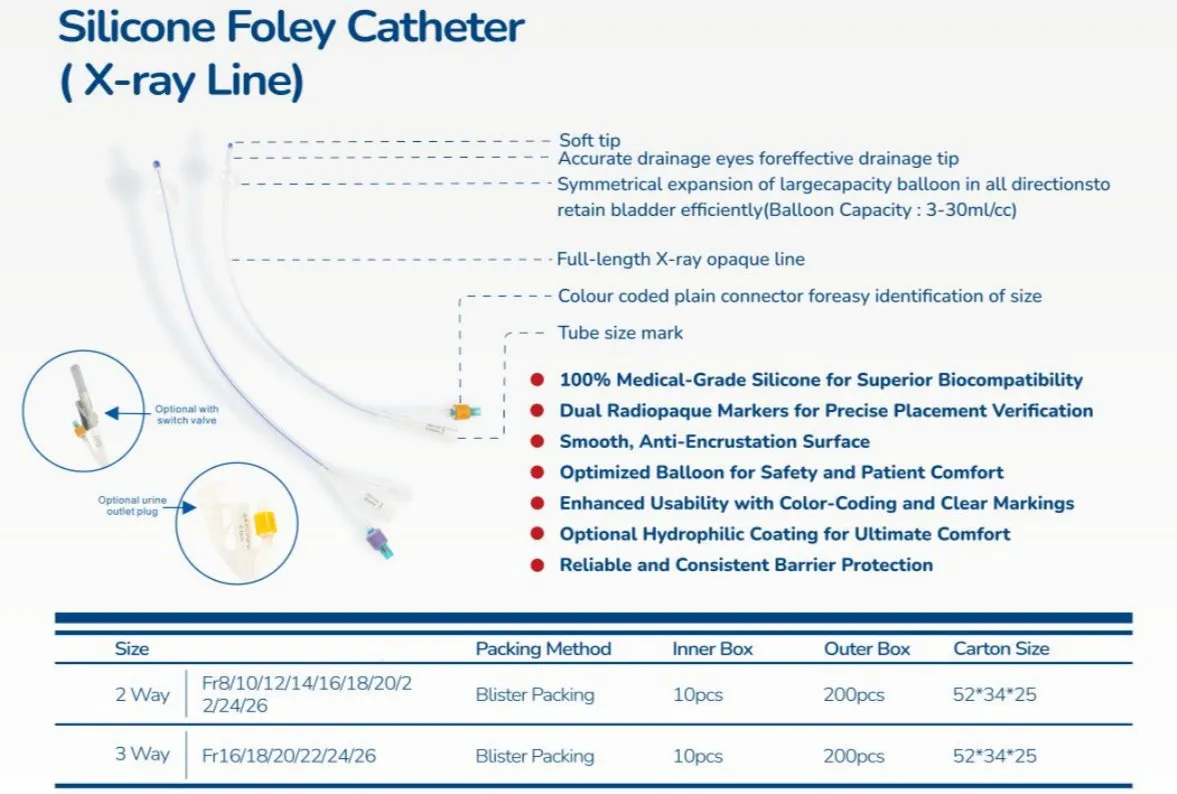



| Catheter Types & Sizes | 2-way: fr8, fr10, fr12, fr14, fr16, fr18, fr20, fr22, fr24, fr26 3-way: fr8, fr10, fr12, fr14, fr16, fr18, fr20, fr22, fr24, fr26 |
| Balloon Capacity | 3-30ml (cc) (adapts to diverse patient bladder retention needs) |
| Packaging | Sterile blister packs; 10 units/inner box, 200 units/outer carton |
| Carton Dimensions | 52x34x25 (optimized for bulk hospital procurement and clinical storage) |
| Core Material | 100% medical grade silicone |





We are a high-tech, innovative healthcare company equipped with robust AI computing power, big data platforms, and affiliations with multiple medical institutes and premium manufacturers. We are dedicated to providing AI-driven digital healthcare services and intelligent solutions for the global healthcare.
Our expertise covers clinical care, rehabilitation, and In Vitro Diagnostics (IVD) fields, with a focus on delivering cutting-edge intelligent solutions. These span surgical procedures, neurosurgical brain-computer interfaces, AI-powered rehabilitation robots, AI-enhanced pathological auxiliary diagnosis, life sciences, as well as comprehensive medical supply chain services (including medical equipment, reagents, and consumables).