
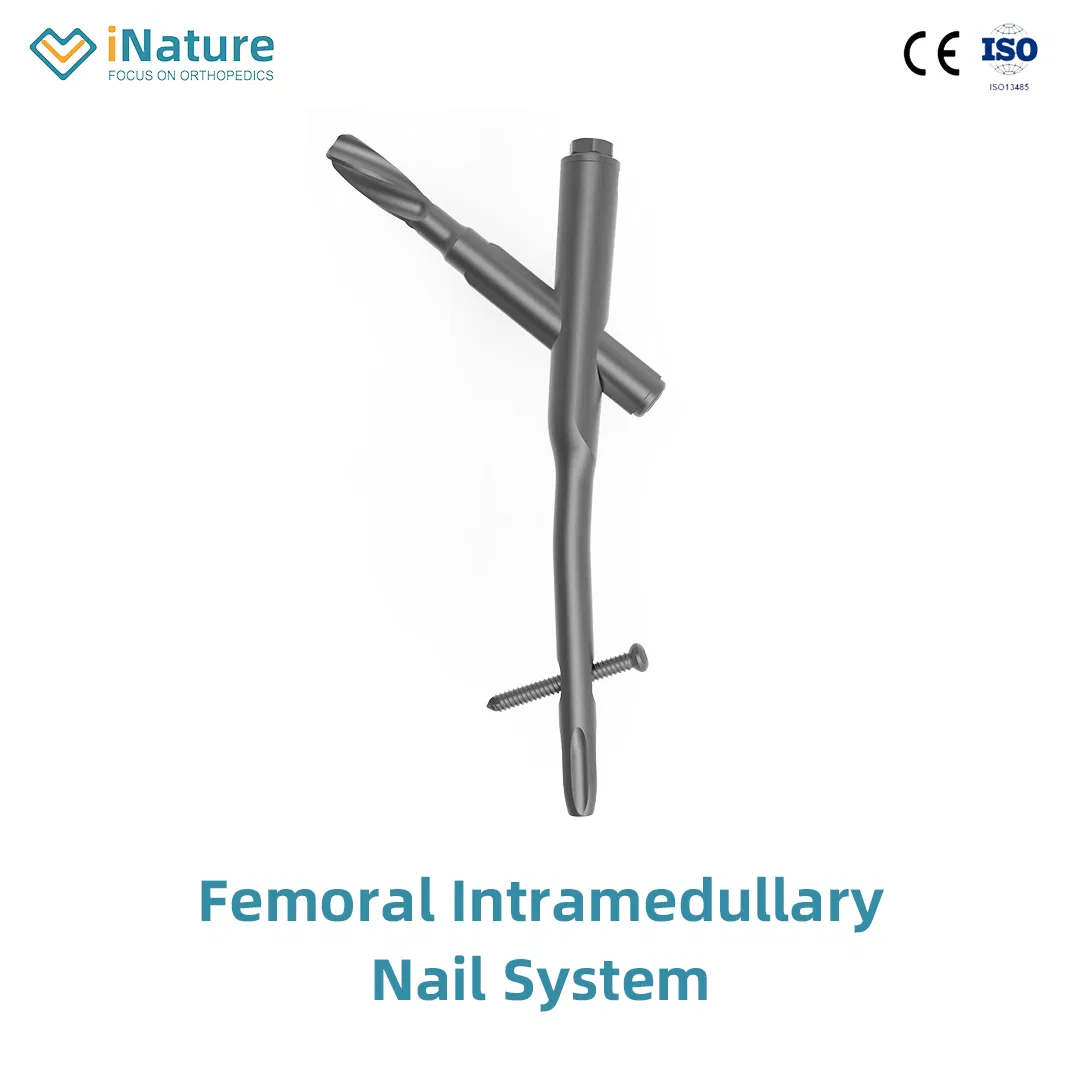



1 / 5
 BRJ Medical
BRJ Medical










| Still deciding? Get samples of $ ! US$ 120/Piece Request Sample |


|
*All the prices are only for reference. Exact price will be based on the latest quotation.



