
1 / 2
 BRJ Medical
BRJ Medical




| Still deciding? Get samples of $ ! US$ 0.1/Piece Request Sample |

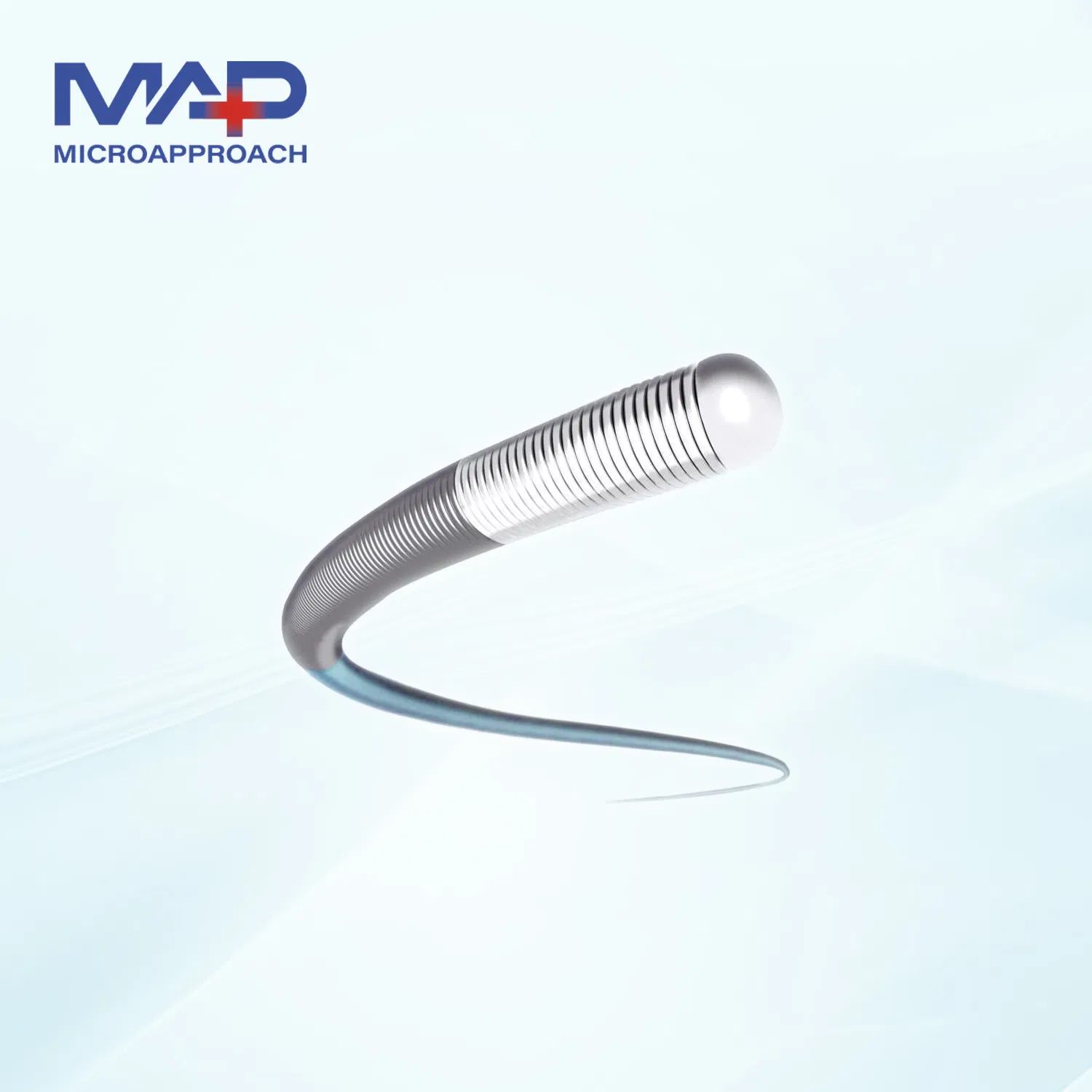
The Silcord PTCA Guidewire is specifically designed to facilitate the placement of balloon dilatation catheters for percutaneous transluminal coronary angioplasty (PTCA) and/or percutaneous transluminal angioplasty (PTA).




| Catalog Number | Diameter (inch/mm) | Length (cm) | Mark Material | Tip Type |
|---|---|---|---|---|
| GGW-14180NA-S | 0.014 / 0.36 | 180 | Gold | Soft |
| GGW-14180NP-S | 180 | Platinum | Soft | |
| GGW-14180NA-XS | 180 | Gold | Extra soft | |
| GGW-14180NP-XS | 180 | Platinum | Extra soft |


