

1 / 5
 BRJ Medical
BRJ Medical










| Customization: | Available |
|---|---|
| Type: | Vacuum Blood Tube & Blood Bag |
| Material: | Borosilicate Glass or Pet Polyester Plastic |

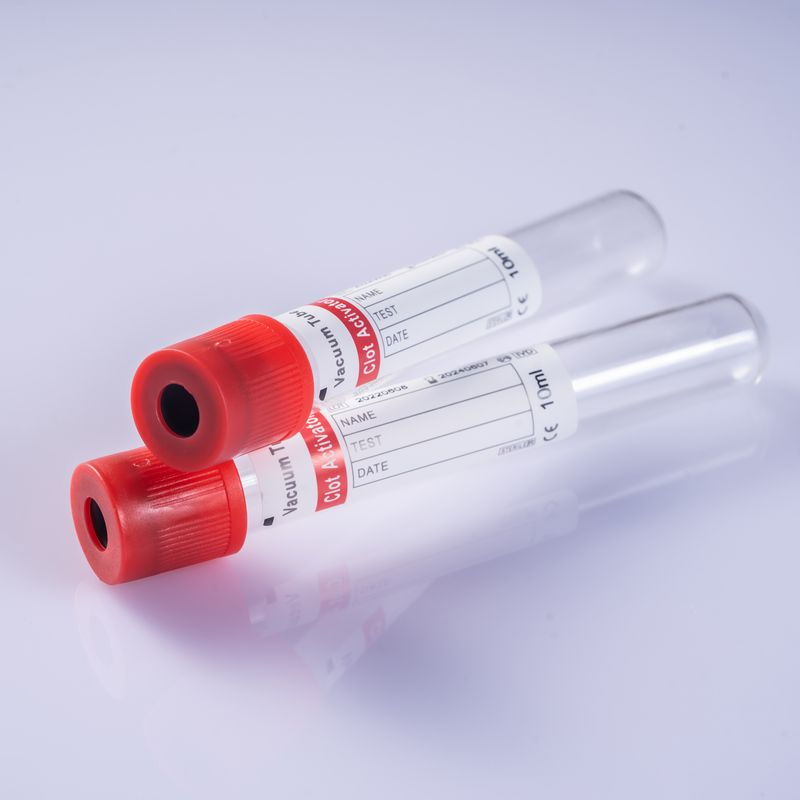
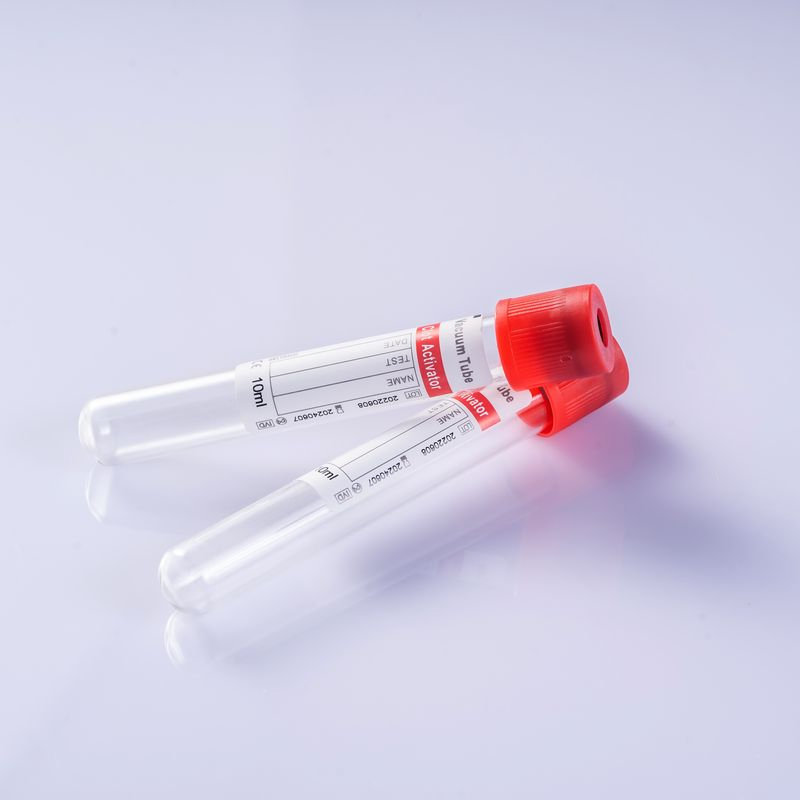
Disposable vacuum blood collection tubes consist of a test tube, rubber stopper, safety cap, additives (anticoagulant or coagulant), separating adhesive, and a label. The tubes are pre-vacuum-sealed and made of borosilicate glass or PET polyester plastic.


Vacuum blood collection tube system guide:
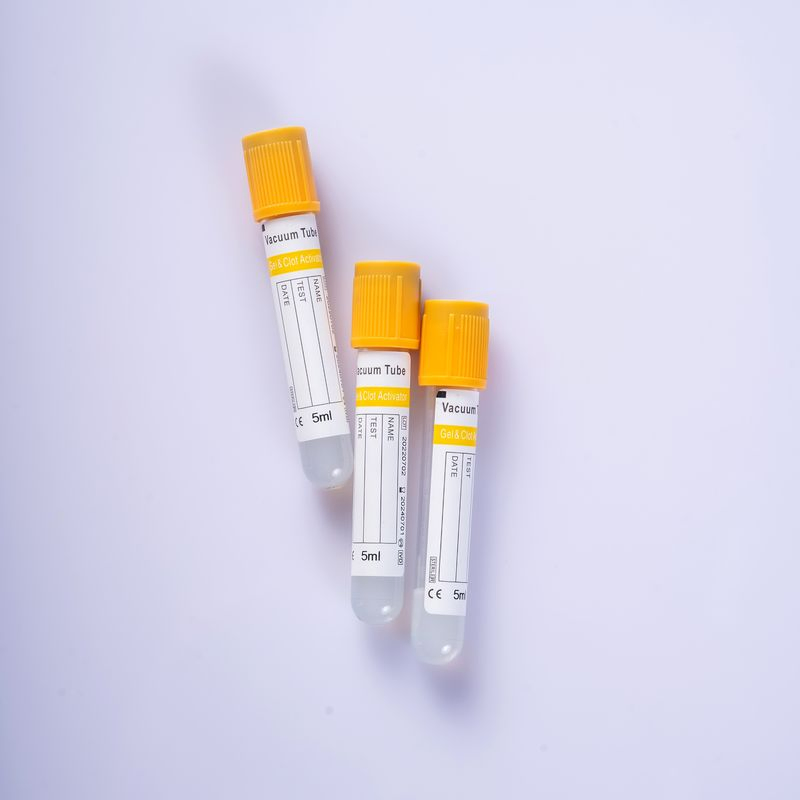
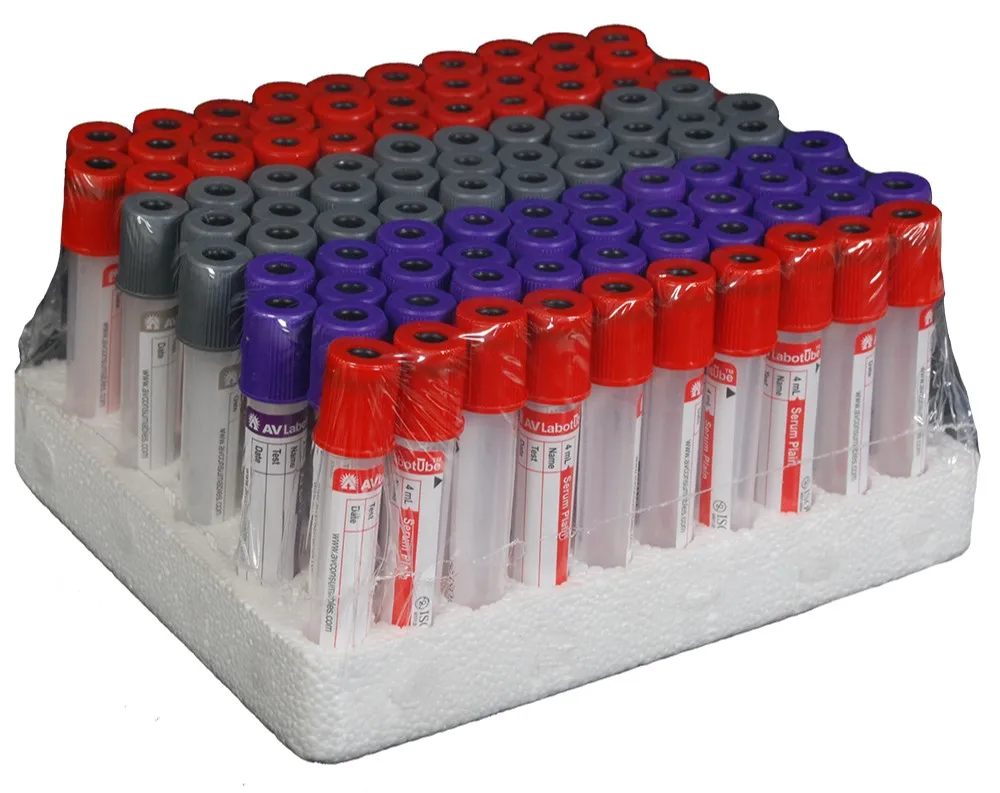
| Material | Cap Color | Usage | Shelf Life |
|---|---|---|---|
| PET / Glass | Orange/Red/Yellow/Purple/Black/Blue/Green/Grey | Hematology Determination, blood type verification | 2 Years (Glass) / 1 Year (PET) |






Founded in 2014, our biological group focuses on the R&D and manufacture of IVD equipment and reagents. With a core team deeply rooted in the market for over 20 years, we cooperate with famous scientific research institutes to set up integrated systems for clinical and diagnostic products.
