


1 / 5
 BRJ Medical
BRJ Medical










| Theory: | Pneumatic |
|---|---|
| Type: | Gas Sterilization Equipments |
| Certification: | CE, ISO13485 |


Note: This system can process two batches of cargo within 24 hours.

| Machine Characteristics | |
| Chamber Capacity | 1 to 100 m3 |
| Chamber Type | Rectangular, 1 to 13 pallets (80x120cm) |
| Doors Type | Double doors with pneumatic sliding systems |
| Material | Stainless steel grade 304 or 316 |
| Heating System | Double jacket hot water circulation system |
| Software & Control | |
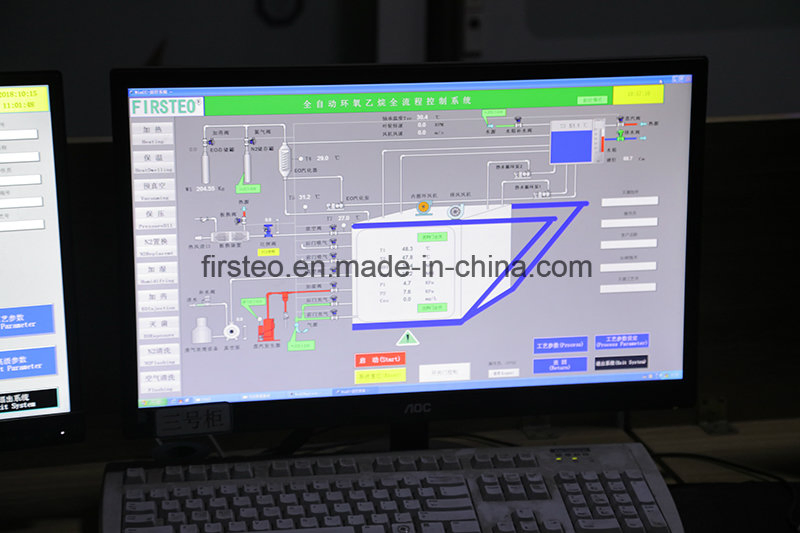
| Control Unit | SIEMENS or SCHNEIDER PLC |
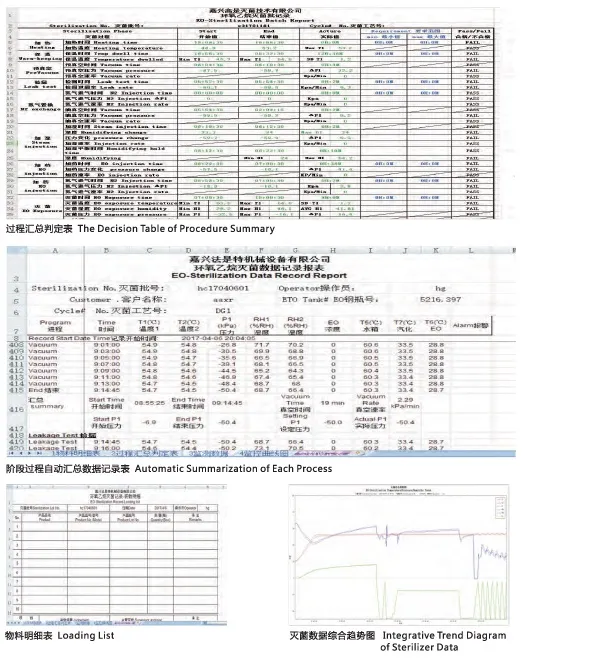
| Features | Automatic/Manual control, Data storage, Graphic recorder |
| Sensors | Precision Pressure, Temperature, and Humidity sensors |
| System Options | |
| Safety | EX/ATEX explosion-proof sensors |
| Gas Support | Nitrogen generator integration |
| Compliance | Software qualified for FDA 21CFR part 11 |