 BRJ Medical
BRJ Medical
 BRJ Medical
BRJ Medical
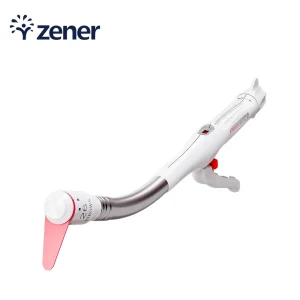 Disposable Medical Surgical Instrument Minimally Invasive System Manual III Rows Circular Curved Cutter Stapler
Disposable Medical Surgical Instrument Minimally Invasive System Manual III Rows Circular Curved Cutter Stapler
 Disposable Linear Cutter Stapler with Reload Units for Open Surgical
Disposable Linear Cutter Stapler with Reload Units for Open Surgical
 Medical Disposable Urinary System Surgical Instrument Skin Linear Cutter Stapling Cutting Electric Curved 30 Stapler
Medical Disposable Urinary System Surgical Instrument Skin Linear Cutter Stapling Cutting Electric Curved 30 Stapler
 Medical Disposable 35W Surgical Staplers Skin Suture Stapler and Staple Remover
Medical Disposable 35W Surgical Staplers Skin Suture Stapler and Staple Remover
Norway’s medical device market is characterized by a sophisticated infrastructure, largely driven by the public health system, Helse Sør-Øst, and other regional health authorities. While the title focuses on Surgical & Examination Gloves Factories, the industrial core of Norway's healthcare procurement has shifted rapidly towards high-precision instruments like Surgical Staplers and Removers.
The demand for minimally invasive surgery (MIS) in Oslo, Bergen, and Trondheim has surged, necessitating advanced stapling technology that ensures rapid tissue healing and reduced post-operative complications. Norwegian medical facilities are among the world's most demanding regarding quality standards and environmental sustainability, often favoring suppliers who provide CE-marked, ISO-certified, and biocompatible instruments.

In Norway, the transition from manual to electric surgical staplers is a defining trend. Surgeons in specialized clinics are increasingly using powered endoscopic staplers to ensure consistent firing tension across varying tissue thicknesses, particularly in bariatric and thoracic cases.
Norwegian "Grønt Sykehus" (Green Hospital) initiatives are pushing factories to innovate in disposable instrument recycling. While instruments remain disposable for sterility, the materials used—such as those in our Biocompatibility Endoscopic Staplers—are being optimized for a smaller carbon footprint.
The use of 35W Skin Staplers in emergency departments across the Nordic region has seen a 15% year-on-year growth. These tools provide a faster alternative to traditional sutures in high-pressure trauma scenarios common in Norway's offshore and remote medical stations.
As robotic-assisted surgery becomes the standard at Oslo University Hospital, the compatibility of stapler cartridges with robotic arms is a key area of R&D for manufacturers targeting the Norwegian market.
Founded in May 2004, Jiangsu BRJ Medical Co., Ltd. is a leading manufacturer specializing in the research, development, production, and distribution of high-quality surgical sutures, surgical needles, and other related medical instruments. Our reach extends globally, with a specific focus on meeting the rigorous demands of the Northern European markets, including Norway.
We are committed to providing a wide range of products that meet the highest quality standards both domestically and internationally. Our products comply with major certifications including CE, ISO 13485, ISO 14001, ISO 45001, and we hold all the necessary production licenses to meet global market requirements. This ensures that every product we manufacture adheres to stringent safety, reliability, and effectiveness standards required by Norwegian healthcare providers.
